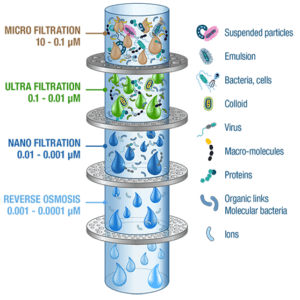
. Occurrence
Chloride, in the form of chloride (Cl−) ion, is one of the major inorganic anions in water and wastewater. The salty taste produced by chloride concentrations is variable and dependent on the chemical composition of water. Some waters containing 250 mg Cl−/L may have a detectable salty taste if the cation is sodium. On the other hand, the typical salty taste may be absent in waters containing as much as 1000 mg/L when the predominant cations are calcium and magnesium.
The chloride concentration is higher in wastewater than in raw water because sodium chloride (NaCl) is a common article of diet and passes unchanged through the digestive system. Along the sea coast, chloride may be present in high concentrations because of leakage of salt water into the sewerage system. It also may be increased by industrial processes.
A high chloride content may harm metallic pipes and structures, as well as growing plants.
- Selection of Method
Six methods are presented for the determination of chloride. Because the first two are similar in most respects, selection is largely a matter of personal preference. The argentometric method (4500-Cl−.B) is suitable for use in relatively clear waters when 0.15 to 10 mg Cl− are present in the portion titrated. The endpoint of the mercuric nitrate method (4500-Cl−.C) is easier to detect. The potentiometric method (4500-Cl−.D) is suitable for colored or turbid samples in which color-indicated endpoints might be difficult to observe. The potentiometric method can be used without a pretreatment step for samples containing ferric ions (if not present in an amount greater than the chloride concentration), chromic, phosphate, and ferrous and other heavy-metal ions. The ferricyanide method (4500-Cl−.E) is an automated technique. Flow injection analysis (4500-Cl−.G), an automated colorimetric technique, is useful for analyzing large numbers of samples. Preferably determine chloride by ion chromatography (Section 4110). Chloride also can be determined by the capillary ion electrophoresis method (Section 4140). Methods 4500-Cl−.C and G in which mercury, a highly toxic reagent, is used require special disposal practices to avoid improper sewage discharges. Follow appropriate regulatory procedures (see Section 1090).
- Sampling and Storage
Collect representative samples in clean, chemically resistant glass or plastic bottles. The maximum sample portion required is 100 mL. No special preservative is necessary if the sample is to be stored.