Pitting corrosion
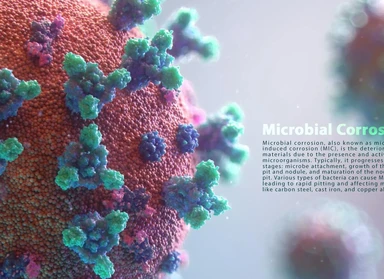
Microbial corrosion based on API 571
Microbial corrosion is a type of corrosion caused by living organisms such as bacteria, algae, or fungi. This type of corrosion is often associated with tubercles or slimy organic materials.

Types of metal corrosion and its classification criteria
The incidence of each type of metal corrosion varies, and the importance of each type will vary across systems and environments. On the other hand, there are striking similarities in the distribution of corrosion damage across similar industries, and by comparing them, it can be seen that corrosion problems in similar industries are very similar.

Corrosion measurement units
In general, to facilitate the study of corrosion problems and to compare different metals with each other, the best method is to express the speed or rate of the corrosion reaction. Corrosion measurement units are used to express corrosion rates.

Corrosion in marine structures and port facilities
Corrosion is often not uniform along the length of a member and is usually confined to the middle or ends of the member. In the case of piles, corrosion may be severe above the waterline and mild or absent below. This area of maximum corrosion is often further away from the sections of maximum anchorage and shear forces, but rebar may be uniform throughout the member.

Causes of tube corrosion in the economizer of the steam generation system
Corrosion in steam generation systems causes many problems in refineries, gas plants and petrochemical plants. Corrosion in boilers is a function of the concentration of oxygen, carbon dioxide and ammonia and the way these species are controlled, and in some cases is caused by the presence of hydroxide and chloride anions or high concentrations of chelating chemicals in the boiler water.

What is corrosion?
The gradual reduction (decomposition) of metals due to chemical and electrochemical interactions is called corrosion, which causes significant financial losses to industries every year. For example, in water systems, various types of corrosion are defined as an electrochemical process.

Corrosion process
Corrosion of metals is the process in which metals reach their original oxidation numbers. In fact, corrosion of metals is an oxidation-reduction reaction in which the metal is oxidized by its surroundings. Typically, oxygen from the air acts as the oxidant in these processes.